About the Seminar:
Paul S. Cremer, Department of Chemistry, Penn State University, University Park PA 16802
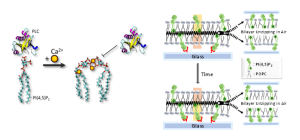
Phosphatidylinositol 4,5-bisphosphate, PI(4,5)P2 is one of the most negatively charged lipids in the plasma membrane of living cells with a net charge ranging from -3 to -5 near physiological pH. Its pendant phosphate groups bind tightly to divalent metal cations including Ca2+ and Mg2+. Herein, we use a variety of spectroscopies and microscopies to investigate the behavior of PI(4,5)P2. For example, it can be shown by vibrational sum frequency spectroscopy that Ca2+ has a far larger influence on the orientation of the inositol ring compared to Mg2+. This is the case because Ca2+ can shed its inner hydration shell to bind directly to the negatively charged phosphate moieties and link them together. Moreover, PI(4,5)P2 has a strong influence on interfacial water structure. Several biological mechanisms involving this lipid will be discussed. These will include its role as a second messenger as well as its ability to aid cytoskeletal attachment.
About the Speaker:
Paul S. Cremer received his Ph.D. in Chemistry in 1996 at the University of California – Berkeley. He then spent two years as the Irving S. Sigal Postdoctoral Fellow at Stanford University before beginning his independent research career in 1998 at Texas A&M University, where he became a distinguished professor and Arthur E. Martell Chair of Chemistry. In early 2013, he moved to Penn State University as the J. Lloyd Huck Chair in Natural Sciences where he currently holds appointments in the Department of Chemistry and the Department of Biochemistry and Molecular Biology. His research is at the crossroads of analytical chemistry, physical chemistry, biochemistry, materials science and engineering. His group has pioneered the use of linear and non-linear vibrational spectroscopies to follow the interactions of ions with peptides, proteins, lipid membranes and macromolecules. This has helped unravel the molecular level mechanisms of the Hofmeister series, a rank ordering of how salt ions affect physical behavior in mixtures of water, organics, and salts. His group has also invented a wide variety of high throughput, low sample volume microfluidic techniques. This includes temperature gradient microfluidics as well as pH modulation sensing, a fluorescence-based technique for detecting the binding of peptides, small molecules, ions, and proteins at lipid membrane interfaces. Most recently, his laboratory has pioneered the study of the metallomembrane, including the tight binding of Cu2+ and other first row transition metal ions to lipids in bilayer membranes containing amine groups such as phosphatidylserine and phosphatidylethanolamine.



